Impurities Training Course - EU edition
15 September - 26 November, 2026
Impurities: from A to Z
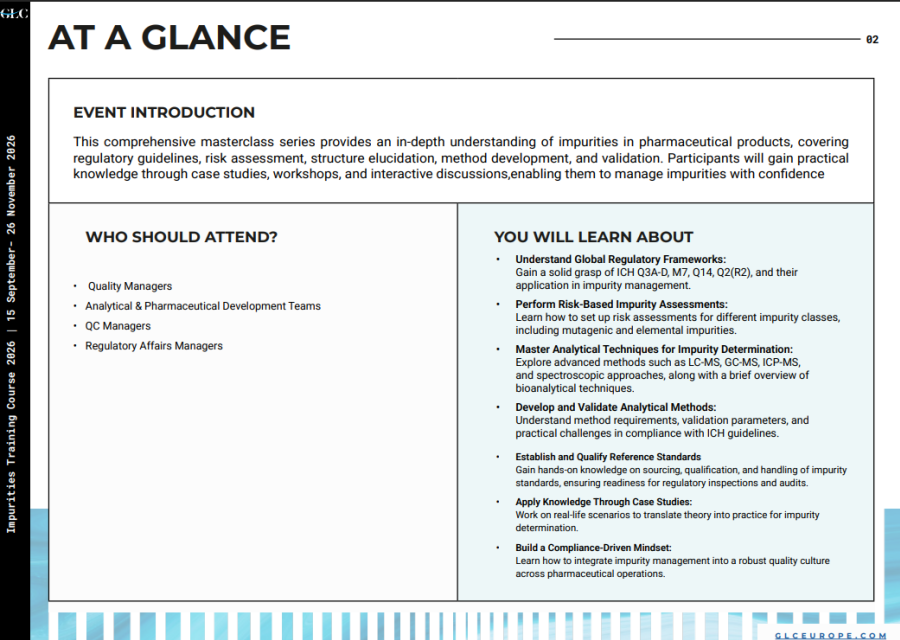
This comprehensive masterclass series provides an in-depth understanding of impurities in pharmaceutical products, covering regulatory guidelines, risk assessment, structure elucidation, method development, and validation.
15 September - 26 November, 2026, Virtual
Time Zone: UTC/GMT+1 /CET
LEARNING OUTCOMES
This comprehensive masterclass series provides an in-depth understanding of impurities in pharmaceutical products, covering regulatory guidelines, risk assessment, structure elucidation, method development, and validation. Participants will gain practical knowledge through case studies, workshops, and interactive discussions, enabling them to manage impurities with confidence.
YOU WILL LEARN ABOUT
-Understand Global Regulatory Frameworks:
Gain a solid grasp of ICH Q3A-D, M7, Q14, Q2(R2), and their application in impurity management.
-Perform Risk-Based Impurity Assessments:
Learn how to set up risk assessments for different impurity classes, including mutagenic and elemental impurities.
-Master Analytical Techniques for Impurity Determination:
Explore advanced methods such as LC-MS, GC-MS, ICP-MS, and spectroscopic approaches, along with a brief overview of bioanalytical techniques.
-Develop and Validate Analytical Methods:
Understand method requirements, validation parameters, and practical challenges in compliance with ICH guidelines.
-Establish and Qualify Reference Standards:
Gain hands-on knowledge on sourcing, qualification, and handling of impurity standards, ensuring readiness for regulatory inspections and audits.
-Apply Knowledge Through Case Studies:
Work on real-life scenarios to translate theory into practice for impurity determination.
-Build a Compliance-Driven Mindset:
Learn how to integrate impurity management into a robust quality culture across pharmaceutical operations
#masterclass #glceurope #pharma #globalleadingconferences #cmc #manufacturing #impurities #referencestandards #methoddevelopment #methodvalidation #leachables #extractables
4 Modules, 12 Days, 4 Key Topics
| TOPIC | DATE | |
| MODULE 1 | Overview Of Impurities And Guidelines, Metal Impurities, Leachables & Extractables, Mutagenic Substances, Residual Solvents, Structure Related Impurities, Risk Assessments | 15-17 September,2026 |
| MODULE 2 | Methods For Structure Elucidation, Establishing And Qualifying Impurity Standards | 07-09 October, 2026 |
| MODULE 3 | Analytical Method Development For Impurity Determination | 03-05 November, 2026 |
| MODULE 4 | Analytical Method Validation For Impurity Determination | 24-26 November, 2026 |
Testimonial
Our success stories
"High attention to detail in course content and very well delivered"
Simon Halsey
Product Development Manager
Essentra Packaging
United Kingdom
Our success stories
"Very good training led by two knowledgeable and open experts. Excellent insight given on many complex topics. Interactive and highly useful"
Aurelie Vivicorsi
USP PD Team Manager
Celonic AG
Switzerland
Our success stories
"Great course, impressed with the knowledge of the trainers and ability to answer wide variety of questions!"
Emilia Szwej
Manager, Senior Investigator
MT Sword Laboratories (BMS)
Germany
About GLC
Global Leadership Conferences began as an ambitious dream by three founders ten years ago. Today its an international series of interactive events, exploring the hottest topics in critical fields. Each year, thousands of professionals join us to challenge the status quo and learn innovative ways to create new solutions in Finance, Pharmaceutical, HR, Health & Safety, and Energy.
know more >14+
Years of experience
600+
Events organized
4,000+
Speakers
25,000+
Attendees

Other events you may like

Training Program for CMC Leaders - EU edition
14th September 2026 - 09th April 2027
Rich with practical insights and real-world applications
learn more >>
Training Program for CMC Leaders - US edition
14th September 2026 - 09th April 2027
Rich with practical insights and real-world applications
learn more >>
Real World Smart Packaging & ePILL Solutions for Pharma MasterClass - US edition
26-27 May 2026
Evolution and progression of smart packaging
learn more >>


























