Free knowledge to monitor the world of events. Have a look at our must read Blogs on Pharma, Finance, HR, Health and Cross Industry.
Connecting everyone with Risks
2021-11-08
Have you ever wondered how to connect everyone with risks? How to capture the attention of the Board to this important issue?

The key is on how you apply the enterprise risk management… and the first step is to involve people of all the job positions when you do the information gathering. Don’t do it as an interview, do it as a conversation and let people know that they are the experts in the process, in the area; that you are there to learn.
When you empower people it’s easier that they are more open to explain in detail what is happening. And this will help you to detect more accurately risks and which controls mitigate them. Even though you may have access to the organization chart, policies, and procedures, you need a person who is doing what the documents say. And after you’ve mapped all the process, show them how it flows, where are the risks and controls. You’ll be surprised by their interest! I’ve lived it several times: the staff wants to know if the activities they do or own, have any risk or if there is a key control.
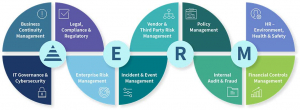
Emerging risks is a tough exercise because we usually can't see what could become a risk. I remember that several years ago, when there was the influenza epidemic, no business included such a catastrophic event in the risk map. When it happened, businesses were closed for several days and that affected sales, causing companies to cut off staff, expenses, benefits. That external risk took companies by surprise and the impact it had was extreme in finance, production, health, and hygiene, etc. Risks can’t be seen, only felt when they are materialized. That's the importance of connecting everyone with risks, so that decision making includes all possible risks that the company may face.

By Mónica Ramírez Chimal - International Speaker & Trainer, Writer, Consultant-Partner at Asserto RSC
Get a feel for our events

Training Program for CMC Leaders - EU edition
14th September 2026 - 09th April 2027
Rich with practical insights and real-world applications
learn more >>
Training Program for CMC Leaders - US edition
14th September 2026 - 09th April 2027
Rich with practical insights and real-world applications
learn more >>
Real World Smart Packaging & ePILL Solutions for Pharma MasterClass - US edition
26-27 May 2026
Evolution and progression of smart packaging
learn more >>













